|
The mass went from 12 to approximately 13. So remember the difference between carbon 12 and carbon 13. Alright by definition, carbon 12 has an atomic mass of 12 A-M-Us and experimentally you can figure out that carbon 13 has a mass of 13 A-M-Us. So this represents 100% of all the carbon atoms. So if you just take these approximate numbers that I have here, so 98.89% of the atoms of carbon in the world are carbon 12, right, and 1.110% of atoms are carbon 13. And so we have two different isotopes for carbon here, so this is carbon 12 and this is carbon 13 and pretty much every carbon atom in the world is one of these two isotopes so I'm not worried about things like carbon 14 because they're extremely small. Now let's look at these numbers here for carbon. Alright, let's do the exact same thing except this time we're going to talk about carbon. 
And so this is the idea of a weighted average. And again, this makes sense because your tests are weighted more than your homework so your final grade is closer to your grade for the test. And to find your grade in the class just add those two numbers together. So point three times 100 and so this is a simple calculation, you can probably do this one in your head. 
So convert the percentage into a fraction so we just move our decimal place two over here so we have point three and then we're going to multiply this by 100. And then let's do the same thing for homework. So point seven times 90 gives you 63 right here. If you're dividing by 100 just move the decimal place one, two, to give you point seven. So all we have to do is move the decimal place. So 70%, so this is a simple calculation, but we have point seven right here so let's go ahead and write point seven. What you need to do first is convert your percentage into a decimal, alright. So what is your grade in the class? So I'm sure some of you guys know how to do this. Let's say that you're a good student and so you average a 90 on all of your tests and you always do all of your homework and so you have 100 in the homework category. And 30% of your overall grade in the class is determined by your homework and so obviously 70 plus 30 gives you 100%. So let's say your teacher weights tests more than homework, so 70% of your overall grade in the class is determined by your test grades. So let's look at this grade distribution here. 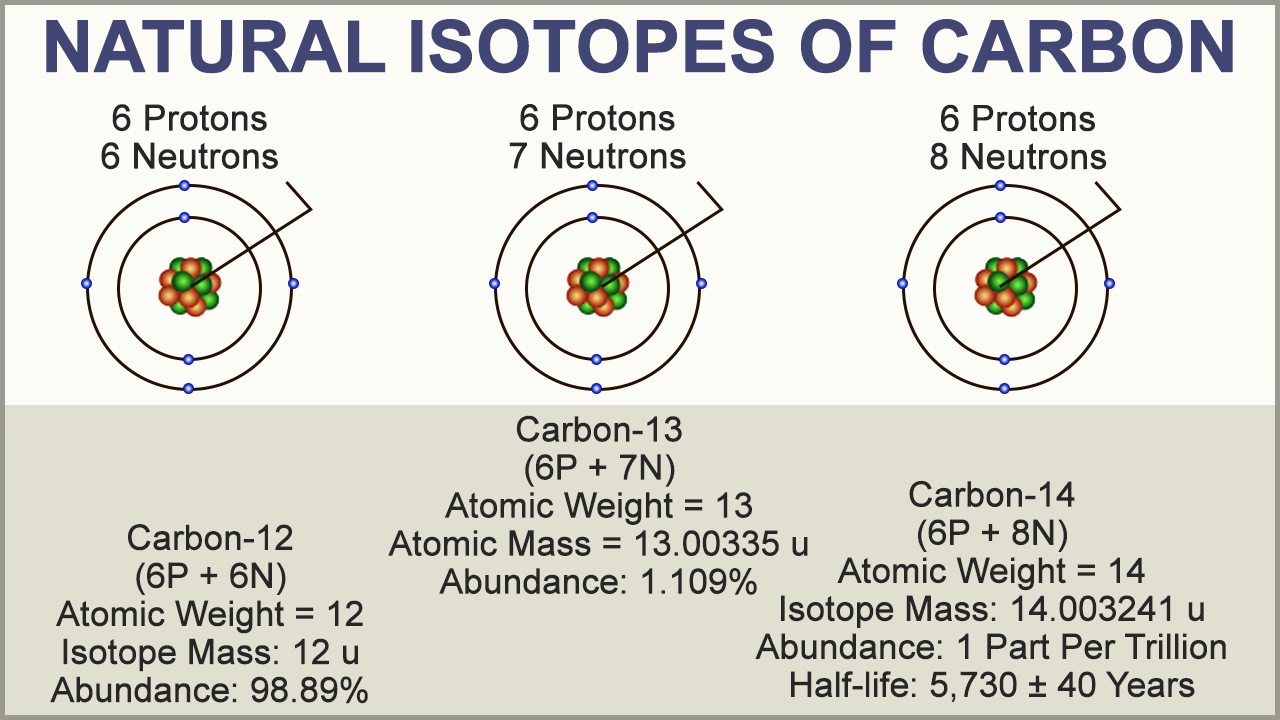
So before we do it for carbon, let's do it for a grade calculation first because this number is actually a weighted average and calculating your grade can sometimes be a weighted average too. And so let's see how we can calculate this number that you'll see on the periodic table for the different elements. So this is just talking about carbon 12, but there are other isotopes of carbon and so the definition for atomic mass is the average mass of all of the isotopes of an element and that's what this number refers to. So this is exactly 12 and this is not, it's 12.01 and so where does the point zero one come from? Well that's because the definition of atomic mass includes the average masses of all the isotopes, alright. This number is exactly 12, 12.00000 and so on. But once again, that number is kind of annoying, it's kind of small, and so it's easier to use this definition of one atom of carbon 12 has a mass of 12 A-M-Us and if you look over here at what you would see on a periodic table, alright, so this number right here is the atomic mass of carbon, but notice it's not exactly 12 A-M-Us. One A-M-U is equal to this many grams and so if you multiplied this number on the right by 12, you'll get the mass of one atom of carbon 12 in grams. So if you wanted to know the mass of a carbon 12 atom in grams, one atom of carbon 12 is equal to 12 A-M-Us. Just to give you an idea of the mass of an A-M-U, so one A-M-U is equal to 1.660, 539 times 10 to the negative 24 grams. The one atom of carbon 12 is equal to 12 A-M-Us and everything else is relative to that value. And chemists took the mass of one atom of carbon 12 as the definition. And since atoms are extremely small, this number would be extremely small and it's annoying to work with small numbers and so instead of working with these extremely small numbers chemists came up with a new term called atomic mass units. 
And normally in chemistry we measure things in grams, so you could just imagine getting a number in grams. So we have this tiny balance that's going to give us the mass of our carbon 12 atom. So just use your imagination here and pretend like you could take one atom of carbon and put it on this tiny little balance here. However, as seen by the helium and sulfur examples, the masses of individual atoms are not whole numbers.- If we wanted to find the mass of one atom of carbon 12, right, one chemistry class, we'd put things on a balance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
